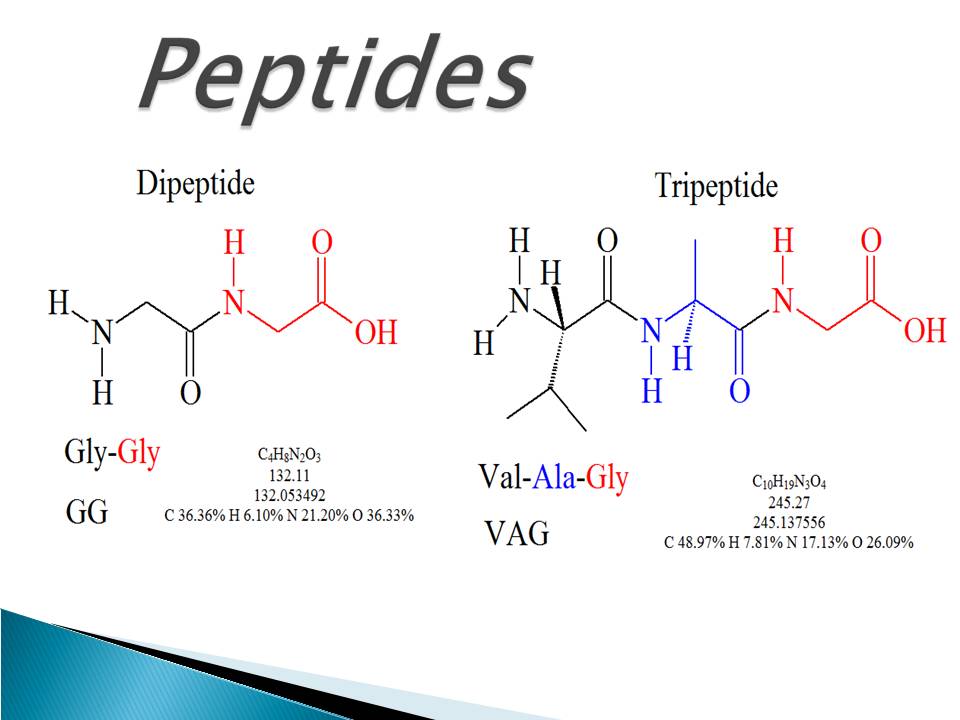
However, each amino acid can twist relative to the next amino acid between the C alpha and C carbons. By "planar" we mean that the 2 alpha carbons, the nitrogen, and carbon and oxygen associated with the peptide bond all lie in a single plane (the peptide bond is resistant to twisting). The shaded quadrilateral represents planar nature of this bond. The peptide bond between two amino acids is depicted. The primary structure is coded for in the DNA, a process you will learn about in the Transcription and Translation modules. The linear sequence of amino acids in the polypeptide chain are held together by peptide bonds and result in the N-C-C-N-C-C patterned backbone. The unique sequence of amino acids in a polypeptide chain is its primary structure. The carboxyl group of one amino acid and the amino group of the incoming amino acid combine, releasing a molecule of water and creating the peptide bond. Each amino acid is attached to another amino acid by a covalent bond, known as a peptide bond, which is formed by a dehydration synthesis (= condensation) reaction. The sequence and the number of amino acids ultimately determine the protein's shape, size, and function. The pattern you are looking for is: N-C-C-N-C-C Try finding the backbone in the dipeptide formed from this reaction. In the process, a molecule of water is released and a peptide bond is formed. The carboxyl group of the first amino acid is linked to the amino group of the second incoming amino acid. Peptide bond formation is a condensation reaction. When we write the sequence of a protein, we will always write it from the from "N to C". When looking at a chain of amino acids it is always helpful to first orient yourself by finding this backbone pattern starting from the N terminus to the C terminus. The amino acids are arranged in a single line- there are no branches. Where the first ("alpha") C will always carry the R group and the second will have a double (ketone) bond to oxygen. There are 20 genetically encoded amino acids available to the cell to build in proteins and all of these contain the same core sequence: These will be used to make the peptide bonds between amino acids in a protein (only the amino groups at very beginning (the "N terminus") and the carboxyl group at the very end (the C terminus) will remain in a polypeptide (= protein). 
The name "amino acid" is derived from the fact that all free amino acids contain both an amino group and carboxylic acid group. Those local structures are stabilised by hydrogen bonds and connected by tight turns and loose, flexible loops.\) For example, the proteins in silk have a beta sheet structure. Two or more parallel or anti-parallel adjacent polypeptide chains of beta strand stabilised by hydrogen bonds form a beta sheet. A Beta strand (β-strand) is a stretch of polypeptide chain, typically 3 to 10 amino acids long, with its backbone in an almost fully extended conformation. The other common type of secondary structure is the beta strand. The alpha helix (α-helix) has a right-handed spiral conformation, in which every backbone N-H group donates a hydrogen bond to the backbone C=O group of the amino acid four residues before it in the sequence. There are two common types of secondary structure (Figure 11). Secondary structure refers to regular, local structure of the protein backbone, stabilised by intramolecular and sometimes intermolecular hydrogen bonding of amide groups. Protein structures are also classified by their secondary structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
